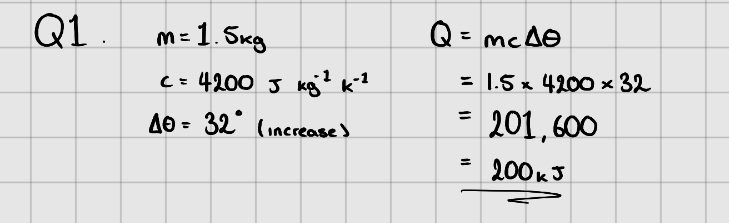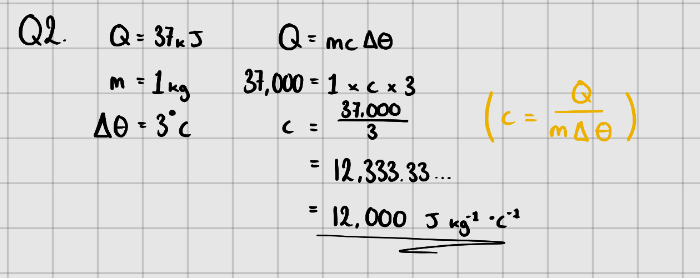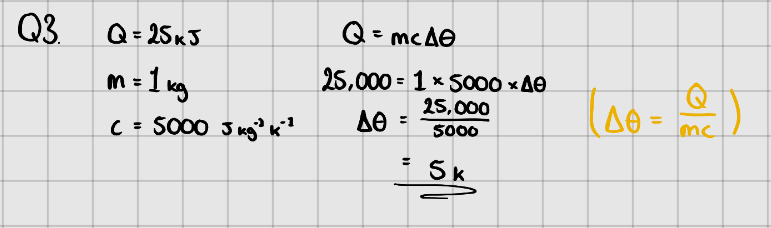Specific Heat Capacity
Specific Heat Capacity - The amount of energy needed to increase the temperature of 1kg of a (pure) substance by $1^{\circ}C$ or $1\space K$.
Equation:
$$ \LARGE{Q = mc\Delta{\theta}} $$
Where, $Q$ - Change in energy ($J$)
$m$ - Mass of substance (kg)
$c$ - Specific Heat Capacity ($J\space kg^{-1}K^{-1}$ or $J\space kg^{-1}\ ^{\circ}C^{-1}$)
$\Delta{\theta}$ - Temperature change in $K$ or $^{\circ} C$